The Ultimate Training to Master Medical Device Quality Engineering & Compliance
Achieve spotless FDA and Notified-Body inspections without hiring pricey external consultants.
Design, Implement, Audit, and Continuously Improve a World-Class Medical-Device Quality Management System
CLICK BELOW TO WATCH FIRST!

4.9/5 star reviews
Thousands of happy customers worldwide
AS SEEN ON
Discover How Bullet-Proof Quality Systems Prevent Recalls, 483s, and Lost Revenue
Does this sound like you?
Uncertain how to interpret new EU MDR and IVDR requirements.
Drowning in documentation with no clear structure or templates.
Fear of costly FDA 483s, warning letters, or product recalls.
Process validations keep failing, delaying launches by months.
CAPA backlog grows while audits reveal repeat findings.
Implement a turnkey QMS using our ready-to-use SOPs and forms.
Run ISO 14971 risk management seamlessly across the product lifecycle.
Execute process validations right the first time and document them flawlessly.
Achieve audit-readiness year-round with a living compliance calendar.
Earn a respected certification that propels your quality-engineering career.
What You Receive in This Program

Fundamentals of Medical-Device Regulations
Map the global regulatory landscape—FDA, EU MDR/IVDR, ISO 13485, and MDSAP—so you know exactly which rules apply.

Risk Management & ISO 14971 Mastery
Apply ISO 14971 risk-management principles from concept through post-market to keep patients—and your company—safe.

Design Controls & DHF Excellence
Create bullet-proof Design History Files and Technical Documentation that sail through audits.

Process Verification & Validation
Validate manufacturing processes the first time using statistics, DOEs, and rock-solid protocols.

Supplier Quality Management
Qualify, monitor, and partner with suppliers to prevent incoming defects and recalls.

CAPA & NC Handling
Turn problems into improvements with a data-driven CAPA and non-conformance system auditors will love.
TESTIMONIALS
What our students are saying...

" We passed our FDA inspection with ZERO 483s just eight weeks after finishing the course. The templates alone were worth the investment! "
- Lisa Chen, QA Manager


" As a start-up we couldn’t afford a full-time RA/QA consultant. This program gave us everything we needed to build a compliant QMS ourselves. "
- John Doe


" I went from junior quality engineer to site quality lead in under a year. The instructor’s mentoring and real-world stories were invaluable. "
- Roberta Johnson

MODULES
FOLLOW MY STEP BY STEP VIDEO TRAINING
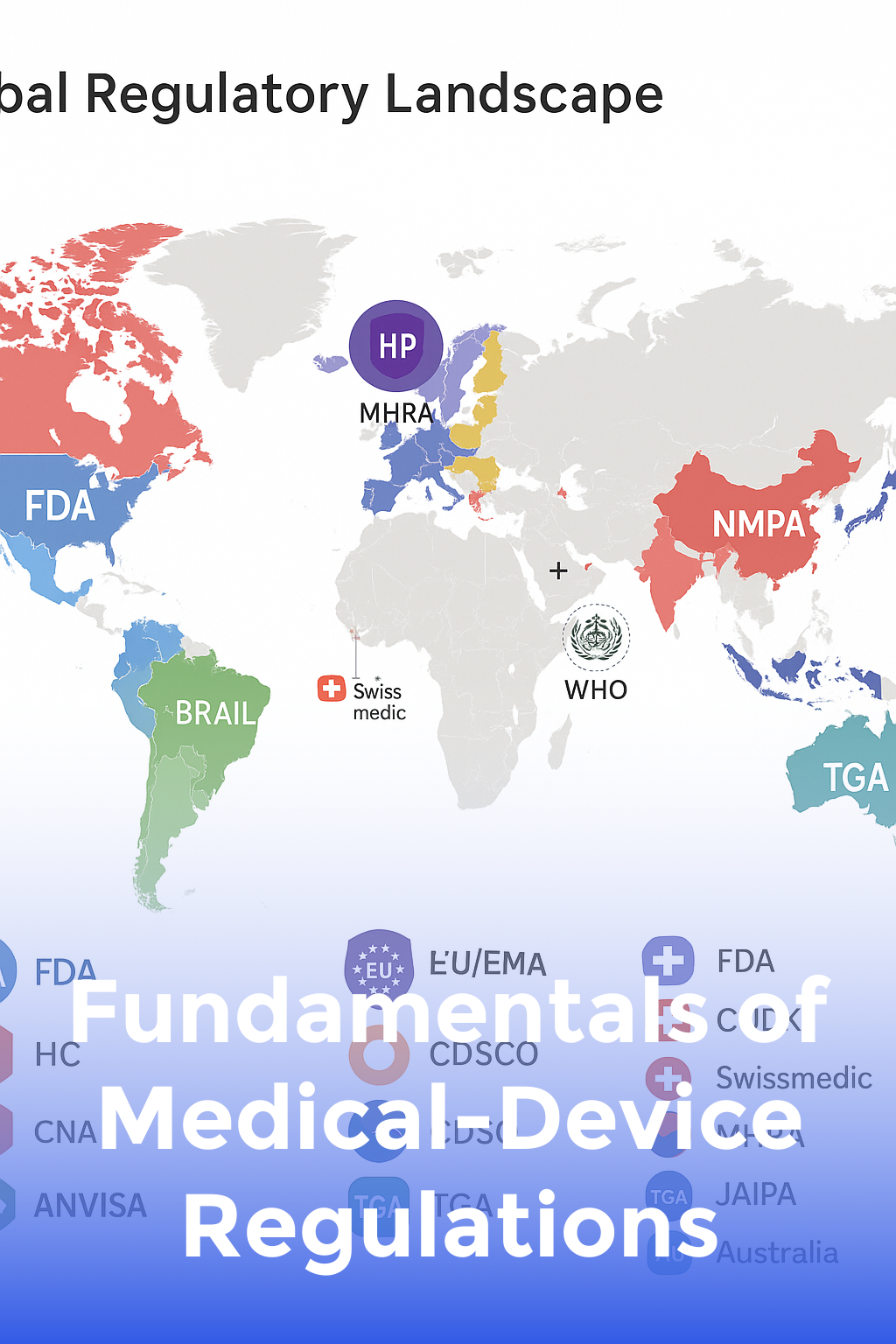
Fundamentals of Medical-Device Regulations
Map the global regulatory landscape—FDA, EU MDR/IVDR, ISO 13485, and MDSAP—so you know exactly which rules apply.
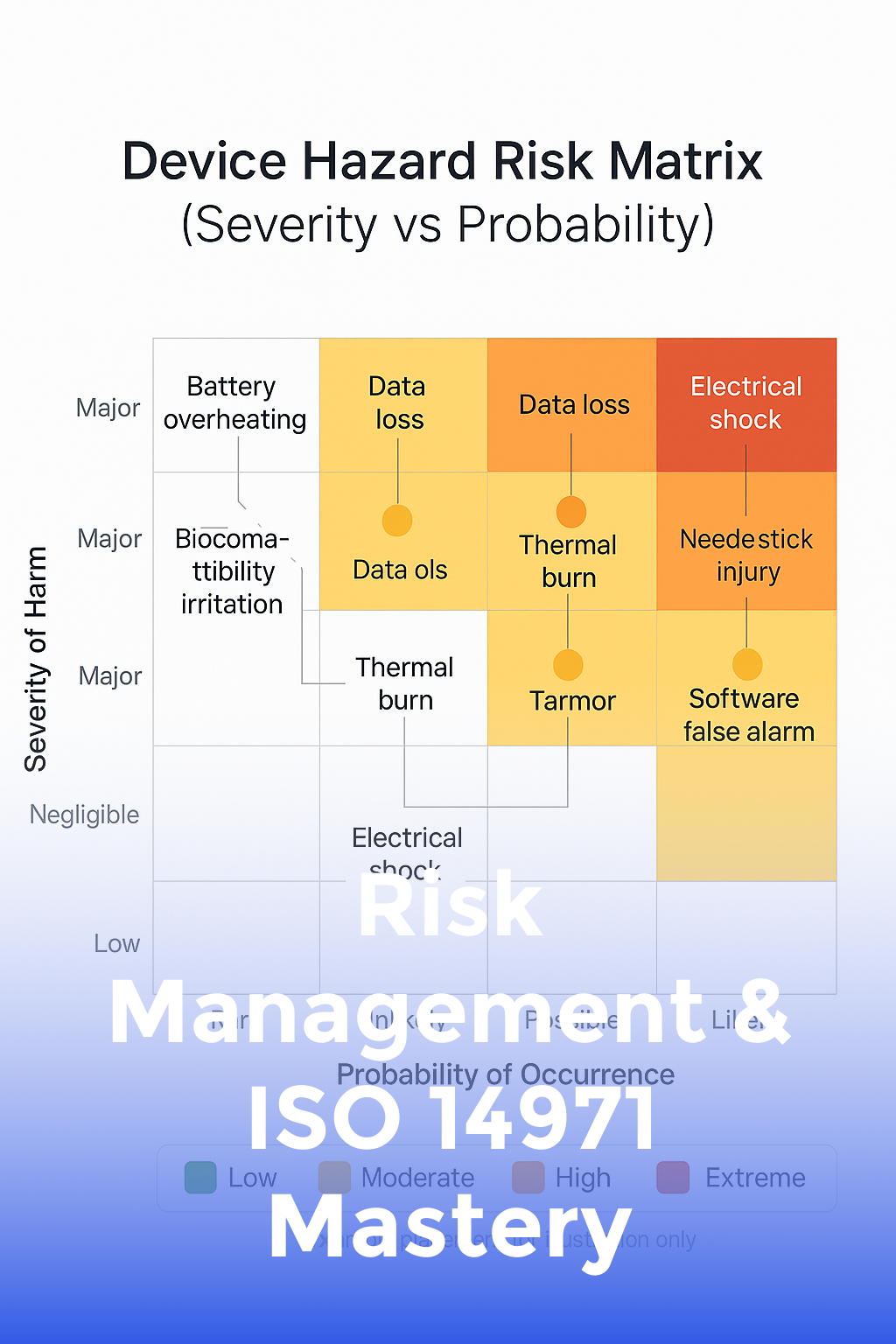
Risk Management & ISO 14971 Mastery
Apply ISO 14971 risk-management principles from concept through post-market to keep patients—and your company—safe.

Design Controls & DHF Excellence
Create bullet-proof Design History Files and Technical Documentation that sail through audits.

Process Verification & Validation
Validate manufacturing processes the first time using statistics, DOEs, and rock-solid protocols.

Supplier Quality Management
Qualify, monitor, and partner with suppliers to prevent incoming defects and recalls.
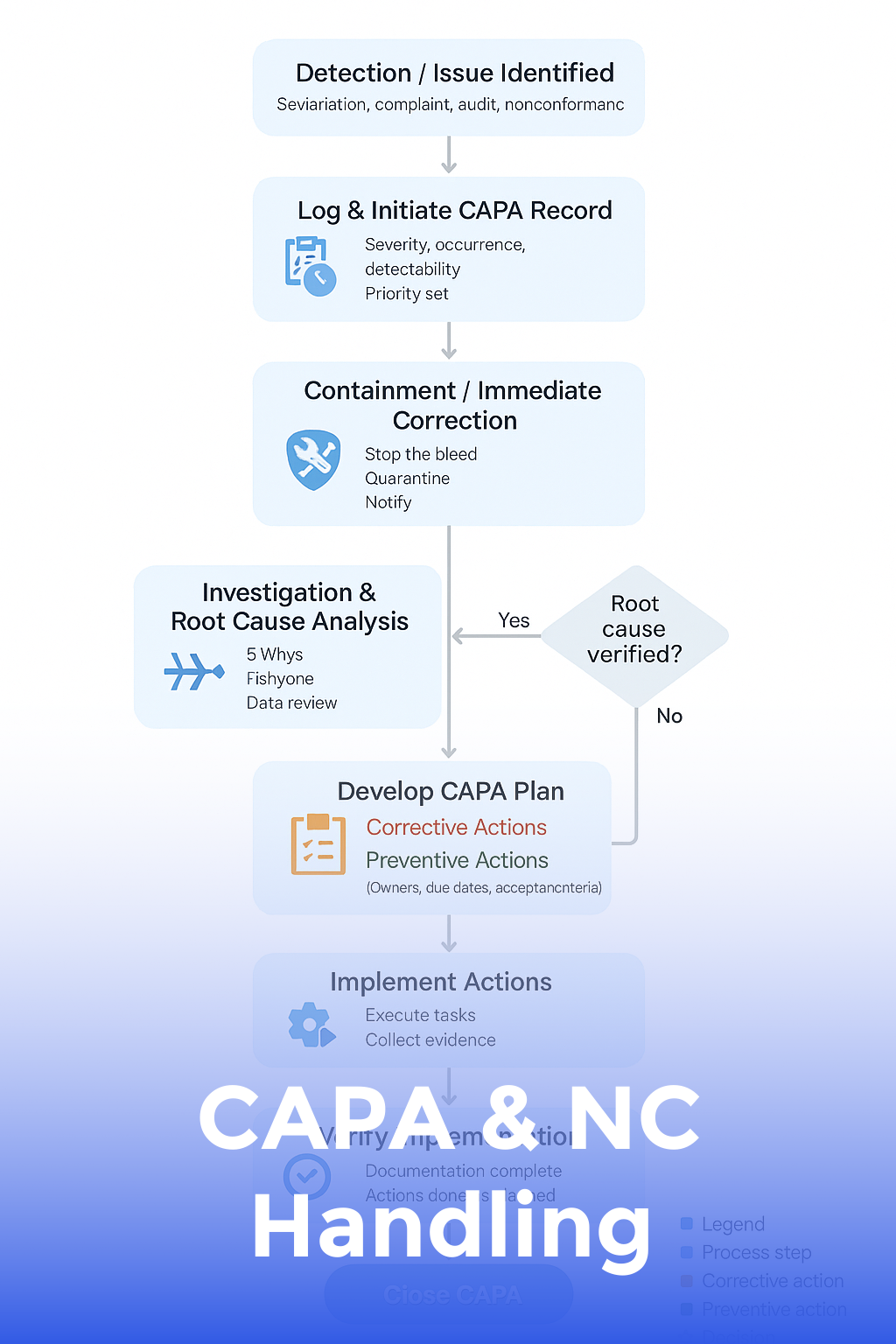
CAPA & NC Handling
Turn problems into improvements with a data-driven CAPA and non-conformance system auditors will love.

Internal & External Auditing Skills
Conduct internal and external audits that uncover gaps before the regulators do.
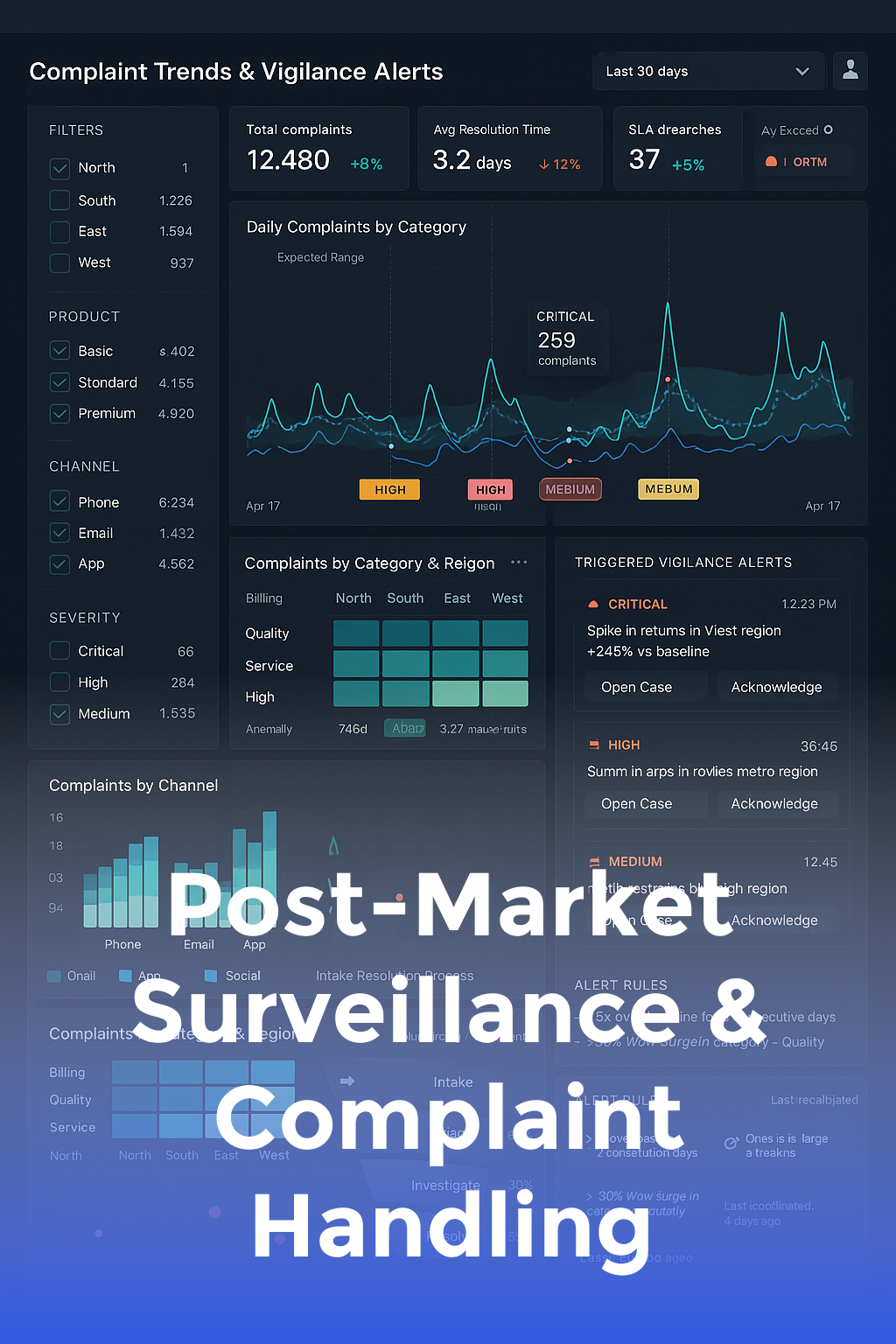
Post-Market Surveillance & Complaint Handling
Build post-market surveillance, complaint handling, and vigilance reporting that close the loop on product safety.

4.9/5 star reviews
Become a Certified Medical Device Quality Engineering Pro
A step-by-step video curriculum and mentoring program that turns regulatory complexity into a clear, repeatable quality system for any medical-device manufacturer.
Here's what you get:
Instant streaming access to 40+ HD training videos, transcripts, and slide decks.
Downloadable SOPs, forms, and templates (ISO 13485 QMS, risk files, validation protocols).
Monthly live group mentoring calls and a private Slack community for Q&A.
Lifetime updates reflecting new FDA guidance, EU MDR amendments, and standard revisions.
Today Just
$997 one time

"Best purchase ever!"
" As a start-up we couldn’t afford a full-time RA/QA consultant. This program gave us everything we needed to build a compliant QMS ourselves. "
ABOUT YOUR INSTRUCTOR
Meet Robert
Your instructor is a 30-year veteran of medical-device engineering, quality, and regulatory affairs. He has designed, validated, and audited Class I, II, and III devices on five continents and helped start-ups and Fortune-500 firms alike sail through FDA and Notified Body inspections.
After seeing the same quality-system mistakes cost companies millions in recalls and inspection findings, he resolved to create a practical, template-driven course that teaches engineers how to build compliant, inspection-ready systems from day one.
Graduates consistently pass FDA, MDSAP, and Notified Body audits with zero 483s or major non-conformances, cut time-to-market by 40 %, and advance to senior or director-level roles within 12 months.
Lead auditor for ISO 13485, MDSAP, and EU MDR with 500+ audits completed.
Guided 100+ successful 510(k), PMA, and CE-mark submissions.
Served on AAMI and ISO technical committees shaping global standards.
Speaker at MedTech Summit, RAPS Convergence, and multiple FDA workshops.
Saved clients over $25 M by eliminating CAPA backlogs and preventing recalls.
Mentored more than 2,000 engineers who now lead quality teams worldwide.

WHO IS THIS FOR...
Perfect for quality engineers, regulatory specialists, auditors, design & manufacturing engineers, and managers within the medical-device industry.
Quality Engineers
Regulatory Affairs Specialists
Design Engineers
Manufacturing Engineers
Supplier Quality Professionals
QA/RA Managers & Directors
Internal & Lead Auditors
Medical-Device Consultants
STILL NOT SURE?
Satisfaction guaranteed
We want you to find value in our trainings! We offer full refunds within 30 days. With all of our valuable video training, we are confident you WILL love it!

STILL GOT QUESTIONS?
Frequently Asked Questions
Do you cover EU MDR, IVDR, and other global regulations?
Absolutely. Every module cross-references FDA, EU MDR/IVDR, ISO 13485, and MDSAP so you can comply worldwide.
What experience level is required?
The program starts with the basics for new quality engineers and quickly scales to advanced topics valuable for seasoned professionals and directors.
How is the mentoring delivered?
You receive monthly live group calls, on-demand replays, and 12-month email access for personalized feedback on your documents.
Enroll in the course now!
Copyrights 2024 | MedDevice Quality Academy™ | Terms & Conditions